DNA nano-chips to capture tumor cells
Detecting metastatic cancer cells circulating in blood vessels is a very difficult task. It involves identifying a serial killer hidden among billions of individuals, diluted in a few drops of blood. The criminal’s fingerprints are certain proteins found on the surface of the circulating tumor cell. Fortunately, small DNA sequences called aptamers are able to recognize them uniquely, even in minute quantities. We have designed nanochips that align thousands of these « police » aptamers, which can stick to tumor proteins in a liquid biopsy and identify the killer cell among millions of normal cells, with a characteristic electrical signal.
One of the main causes of death for cancer patients is organ failure induced by metastasis development. The primary tumor spreads through the body by generating the circulating tumor cells (CTCs), that is cancer cells that detach from the tumor and circulate within the blood vessels. Our institute IEMN is partner in an international, multidisciplinary research effort to develop an innovative, original family of lab-on-chip devices, aimed at detecting CTCs with high efficiency. The devices are based on a sophisticated quantum-mechanical effect, which allows to detect the presence of a cancer cell immobilized on the chip with extreme precision. In short, a DNA fragment carrying a redox molecule on one end is sandwiched between two electrodes; at equilibrium, the DNA fluctuates randomly and the redox molecule induces a very weak current, via the quantum tunneling of electrons from/to the negative electrode; when a CTC is present, the DNA is able to identify the presence of cancer-specific antigens found at the surface of CTCs, whose presence is detected by the change in the quantum tunneling current. The first results of this technique have been recently published in the journals Biosensors and Bioelectronics and ACS Sensors. In the following, we give a more complete description of the technique and its main results.
The first protagonist character of this story is the epithelial cell-adhesion molecule (EpCAM). This protein, found at the outer membrane of epithelial cells, has received a vast attention as the main membrane marker used to isolate CTCs. The biology of EpCAM and its role are not completely understood, but evidence suggests that the expression of this epithelial cell-surface protein is crucial for metastatic CTCs, since most cancers originate from epithelial cells. A typical clinical screening result is that the progression-free survival and overall cancer survival times are critically shortened in patients with ≥5 CTCs per blood sample of 7.5 mL, or ≥20 EVs positive to EpCAM. Consider that such volume of blood contains about 40 billion red blood cells and about 50 million other cells (leucocytes, granulocytes), and you can get an idea of the level of resolution that is required, to capture and discriminate a single CTC from the bulk of normal cell mass.
The other main characters of the play are DNA aptamers. Aptamers are artificially synthesized, short, single-stranded fragments of DNA or RNA (ssDNA or ssRNA) that can bind to a specific target protein, as well as to peptides, carbohydrates, small molecules, toxins, and even live cells, with an extreme selectivity down to the nM range (meaning that they can selectively find 1 target element out of a one-billion sample). Since their discovery in the early 1990s, great efforts have been made to make them clinically relevant for diseases like cancer, HIV, and macular degeneration. With advances in high-precision medicine, targeted-therapy, imaging, and nanotechnologies, aptamers are readily considered among the best potential targeting ligands thanks to their low-cost chemical synthesis and ease of modification to adjust to variable targets.
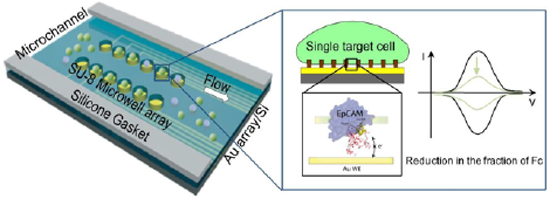
If the DNA aptamers and the EpCAM proteins found at the surface of CTCs are two lovers to be married, here comes the reverend father that conducts the ceremony. It is a variant of the electrochemical method of cyclic voltammetry (CV), a fairly old and standard experimental technique that almost anybody could make work in their garage. In its most basic setup, CV detects the presence of molecules susceptible of transferring electrons, that is redox species that can be reduced or oxidized; by varying periodically the potential of the negative electrode, in such a way to sweep across the energy levels of the redox molecule, electrons can be transferred to the molecule and back, thereby generating a current. The shape of the potential vs. current plot is very typical of each molecular species, and describes the energy barrier that electrons must pass to move between their two stable states. Here we turn the general CV method into an extremely sophisticated, microscopic diagnostic tool. In our experiments, a redox molecule is fixed on one end of a short DNA aptamer, which in turn is fixed on a gold electrode by its other end. In such a configuration, the DNA behaves like a tether that makes the position of the redox molecule to fluctuate randomly above the electrode surface, between a minimum approach distance and a maximum elongation distance. Given such a variable distance from the surface, the probability of electron transfer from and to the electrode is a sharp exponential function: when the molecule fluctuates close to the surface, the electron can pass the barrier by quantum tunneling and give rise to a weak, yet measurable current; but when the molecule is fluctuating away from the surface the current goes to zero. And this is just the key working principle of our innovative CTC detection method: since the DNA aptamer sequence is designed to selectively recognize the EpCAM protein, when a CTC cell is present “above” the electrode in the solution, the DNA aptamer will stick to it, thus blocking the redox species at a position well above the electrode surface, and effectively shutting off the current signal.
The actual device is a microfluidic lab-on chip, with size of a few mm2 (see the figure). The surface of the Au electrode is decorated with a regular array of plastic (reticulated hydrogen silsesquiloxane) nanopillars, 20nm in height and 200nm wide, and spaced by 500nm. The DNA aptamer attached to the Au surface has an extended length of about 15 nm, however at room temperature it adopts a partly-folded conformation, which reduces it to a more compact structure of ~10 nm; a ferrocene molecule (redox species) is attached at the free DNA end. Ferrocene-carrying DNA aptamers are deposited as a dense monolayer on the Au surface. The whole device is built in a microfluidic chip, in which a solution containing various types of target cells is flown at low speed. In the first experiments, CAPAN-2 cells from pancreatic cancer and RAMOS cells from Burkitt lymphoma lines were used as test. Flowing cells are trapped on the nanopillars and remain suspended, slowly dropping towards the surface by gravity. The surface membrane proteins, among which the target EpCAM, can so come within reach of the fluctuating DNA tether. After each DNA gets attached to an EpCAM, this contributes a reduction of the total measured current, in the microampere range. The present lower resolution limit is about 3000 cells per mL, still high compared to the target requirement. However, this first version of the technical setup has large margins for improvement, and is very promising because of its versatility (target proteins can be easily changed by changing the DNA aptamer sequence, different proteins can be simultaneously targeted by using multiple aptamers in parallel), and also because of its very reduced fabrication cost compared to current mainstream technologies.
The team is made up of a multidisciplinary group of physicists, chemists, engineers and biologists from the LIMMS laboratory in Tokyo (Japan), the IEMN and the CANTHER cancer research unit, both in Lille (France). The IEMN team’s contribution focuses on electrode surface chemistry and DNA immobilization, led by Dr. Yannick Coffinier, and on computer simulation of DNA-protein interactions in solution, led by Prof. Fabrizio Cleri .