Toward a more sustainable method for converting methane into hydrogen and hydrocarbons using an electronic phase transition in vanadium dioxide
Optimizing the efficiency of photocatalysts is a key challenge in methane conversion. This process relies on light absorption and the transport of photo-induced charges to the surface of the photocatalysts.
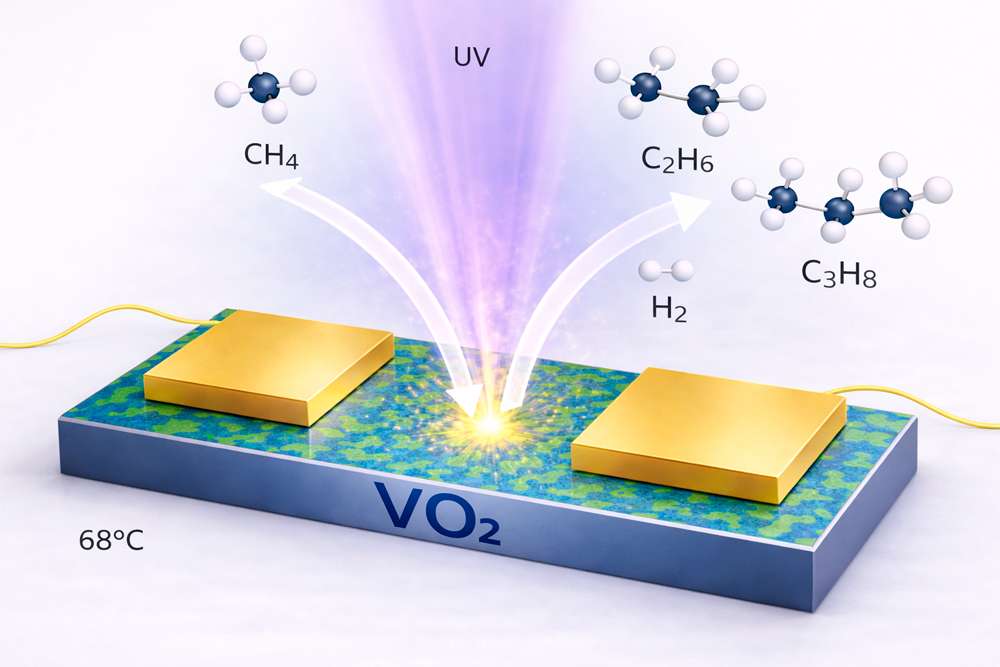
Researchers have recently shown that vanadium dioxide (VO₂), which undergoes a phase transition at 68 °C, significantly facilitates the transfer of these charges to react with methane during this transition.
Thin films of VO₂ thus enable record-breaking photocatalytic conversion of methane into hydrogen, ethane, and propane. By reducing their thickness, the reaction yields a single hydrocarbon—propane—offering a more sustainable method than current industrial processes.
Methane is a very abundant gas but difficult to convert because it is chemically inert. To overcome this issue, researchers use photocatalysts —materials that absorb light energy and convert it to electrical charges that trigger a chemical reaction. However, much of this energy is quickly dissipated, making the activation of methane on the surface of the photocatalysts inefficient.
A team of scientists(1) has shown that a vanadium oxyde compound (VO₂) possesses a property that is ideal for overcoming this problem.
At 68 °C, VO₂ undergoes a sudden phase transition: it changes from an insulating to a metallic state. During this transition, the two states coexist, intermingling on the nanoscale. The numerous boundaries between these regions effectively separate the charges generated by light. These charges then reach the material’s surface in large numbers, making the reaction much more efficient.
Thanks to this transition, thin VO₂ films convert methane into useful molecules such as hydrogen, ethane, or propane, with, under certain experimental conditions, selective production of one of the two hydrocarbons. The transition can even be triggered electrically at lower temperatures, paving the way for more efficient and energy-saving photocatalysts.
(1) IEMN (University of Lille, CNRS, École Centrale Lille, École Polytechnique Hauts-de-France, Junia), UCCS (University of Lille, CNRS, École Centrale Lille, ENSCL, University of Artois), UMET (CNRS, INRAE, École Centrale Lille), LASIRE (University of Lille, CNRS), LPEM (ESPCI Paris, PSL University, CNRS, Sorbonne University), XLIM and IRCER (CNRS/University of Limoges).
Learn more
Contact : bruno.grandidier![]() univ-lille.fr
univ-lille.fr
Double Honor for Ph.D. Candidate My Nghe Tran
As part of these research activities, the Ph.D. candidate My Nghe Tran, supervised by Bruno Grandidier and Vitaly Ordomsky (UCCS-CNRS), has distinguished himself through innovative work in photocatalysis, which was recognized with the University of Lille’s Best Thesis Award. »
On December 4, 2025, My Nghe Tran had already had the honor of receiving the first thesis prize awarded by the French Chemical Society (SCF), Hauts-de-France section.
This award recognizes the significance, originality, and innovative contributions of his doctoral thesis, which focused on improving the efficiency of energy conversion via photocatalysis using a moderate external electric field.